
The Iverson Health Innovation Research Institute’s overarching vision is to identify, understand and solve real-world health challenges across the human lifespan. Providing an evidence base for innovation in personalised healthcare, our Institute strategically cultivates connections both within Australia and across the globe, drawing on existing partnerships and forging new ones from research, industry and government.
Our key capabilities
Bioengineering
Digital Healthcare
Personalised Healthcare
Our research
We exploit Swinburne’s expertise in mental health, neuroscience, ageing, disability, bioengineering, data science, digital technologies, design and social science to pursue our core research programs.

Securing embedded AI systems in AR/VR for health care
This collaborative research project aims to address the complex and crucial need for developing secure AI systems used in health care.
-

How bioengineering tools could help treat severe depressive disorders
For the millions of patients suffering from treatment-resistant depression, new bioengineering research is pointing the way to personalised breakthroughs.
-

Bridging the Dementia Care Gap
e-DiVA - the empowering Dementia Carers with an iSupport Virtual Assistant is a culturally adapted digital support platform designed with and for dementia carers across the Asia-Pacific region.
Our people
Our Institute is named after Professor Don Iverson, the Inaugural Executive Dean of the Faculty of Health, Arts and Design at Swinburne and an internationally acclaimed academic leader and researcher in health and medical sciences.
Our latest news
-

- Technology
- Social Affairs
- Health
- Science
Driving meaningful progress in assisted living tech
Swinburne’s MedTechVic hub annual showcase brings together research, lived experience and practical innovation in assisted living technologies. This year's theme was ‘Real innovation, real lives’, highlighting the hub’s ongoing commitment to driving inclusive innovation.
Thursday 12 February 2026 -
- Health
- Science
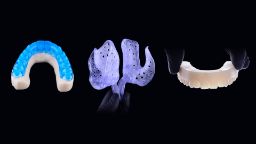
Swinburne to boost AI-driven design for dental devices with investment in H3D
Swinburne has invested in spinout company H3D, a cutting-edge tech company that is reinventing custom-fit devices using AI and automation.
Monday 31 March 2025 -

- Health
- Science
Swinburne to advance cardiac care and asbestos detection with industry-engaged grants
Swinburne has secured two grants from Australia's Economic Accelerator Ignite program to support innovative commercialisation projects.
Wednesday 12 March 2025 -

- Health
- Science
Medical cannabis mysteries to be unlocked by new five-year NHMRC project
A new Swinburne-led medical cannabis investigation aims to help close the knowledge gap surrounding the drug and its potential in society.
Wednesday 26 February 2025 -

- Health
- Science
- University
- Technology
Swinburne’s 2025 MedTechVic showcase celebrates innovation in collaboration
Swinburne University of Technology’s MedTechVic hub brought together researchers, industry partners and end users of medical and assistive devices at their recent 2025 annual showcase.
Thursday 20 February 2025
Find more news articles about the Iverson Health Innovation Research Institute.
Are you interested in joining our team?
Get in touch if you are interested in doing a PhD or a post-doctoral role looking at real-world heath challenges and innovations across the lifespan.
Email ihi@swinburne.edu.au
Our collaborating research centres and hubs
-
Centre for Astrophysics and Supercomputing
-
Centre for Design Innovation
-
Centre for Forensic Behavioural Science
-
Centre for Mental Health and Brain Sciences
-
Centre for Transformative Media Technologies
-
Data for Social Good Cloud Innovation Centre
-
Swinburne Design Factory Melbourne
-
Digital Innovation Lab
-
Digital Capability Research Platform
-
Factory of the Future
-
Swinburne Innovation Studio
Discover our other research institutes
Subscribe to our mailing list
Keep up to date with the latest news and events from the Iverson Health Innovation Research Institute.
By signing up, you consent to MailChimp collecting your data, which is treated in accordance with Swinburne's Privacy Statement. You can unsubscribe at any time.Contact the Iverson Health Innovation Research Institute
If your organisation is dealing with a complex problem that you’d like to collaborate on with us, or you simply want to contact our team, get in touch by calling +61 3 9214 8180 or emailing ihi@swinburne.edu.au.











